Group-A
1. Multiple choice question.
1.1 Which of the following greenhouse gasses has maximum contribution towards global warming?
(a) N2O
(b) CH4
(c) CO2
(d) H2O vapour (Ans)
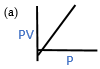
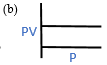
1.2 According to Boyle’s law which is the PV vs P graph?


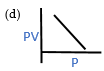
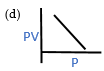
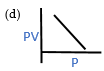
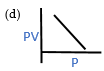
1.3 If the vapour density of a carbon containing gaseous substance is 13, which of the following can be its molecular formula?
(a) CO2
(b) C2H4
(c) C2H6
(d) C2H2 (Ans)
1.4 The unit of coefficient of linear expansion of a solid is
(a) m
(b) M^(-1)
(c) °C^(-1) (Ans)
(d) °C
1.5 An object is placed in between the optical centre and focus of a thin convex lens. What is the nature of image of the object?
(a) real and inverted (Ans)
(b) virtual and inverted
(c) real and erect
(d) Virtual and erect
1.6 When a ray of light is incident perpendicularly on a transparent glass slab, what will be its angle of deviation?
(a) 0° (Ans)
(b) 180°
(c) 30°°
(d) 90°
1.7 Which of the units given below is the SI unit of resistance?
(a) volt
(b) ampere
(c) coulomb
(d) ohm (Ans)
1.8 In domestic electric circuit the fuse wire is connected to which of the following?
(a) earth line
(b) live line (Ans)
(c) neutral line
(d) both live and neutral line
1.9 β-ray emitted from a radioactive element is
(a) a stream of electrons (Ans)
(b) a stream of protons
(c) a stream of neutrons
(d) electromagnetic wave
1.10 How many groups are in the long periodic table
(a) 7
(b) 8
(c) 9
(d) 18 (Ans)
1.11 In formation of which of the following compounds octet rule is not obeyed?
(a) NaCl
(b) LiH (Ans)
(c) KCl
(d) CaO
1.12 Which of the following can conduct electricity?
(a) molten NaCl (Ans)
(b) liquid HCl
(c) solid NaCl
(d) aqueous solution of glucose
1.13 What will be the colour of the resulting solution when excess aqueous ammonia is added to an aqueous solution of copper sulphate?
(a) Yellow
(b) Green
(c) deep blue (Ans)
(d) Brown
1.14 In which of the following alloys zinc is present?
(a) bell metal
(b) Brass (Ans)
(c) Bronze
(d) Duralumin
1.15 Which of the following is a saturated hydrocarbon?
(a) C3H6
(b) C2H4
(c) C2H2
(d)C2H6 (Ans)
Group-B
2. Answer the following question (alternatives are to be noted).
2.1 Mention one use of biogas.
Ans: Domestic cooking and lighting in villages.
Or,What is the role of NO in decomposition of ozone in the ozone layer?
Ans: Several chemicals such as oxides of nitrogen (commonly referred to as NOx), chlorofluorocarbons (CFCs) etc.destroy ozone to create ozone hole.
2.2 Among charcoal, petrol and ethanol which one is a fossil fuel?
Ans:Petrol is one of the fossil fue.
2.3 Under constant pressure at what temperature in degree Celsius the volume of an ideal gas will be 0 according to Charles’ law?
Ans: Under constant pressure at -2730C temperature the volume of an ideal gas will be 0 according to Charles’ law.
2.4 What is the unit of M in the equation PV=W/M RT? (symbol have usual meaning)
Ans: The unit of M is gram in the equation PV=W/M RT.
2.5 Whether the following statement is true or false?
The real expansion of any liquid depends on the expansion of the vessel in which it is kept. False
Or,Among iron , invar and copper which one has the least coefficient of linear expansion?
Ans: Among iron , invar and copper invar has the least coefficient of linear expansion.
2.6 Between the angle of incidence and the angle of refraction which one is greater when light travels from a rarer to a denser medium?
Ans: Angle of incidence will greater when light travels from a rarer to a denser medium.
2.7 What type of mirror is used in the view finder of a motor car?
Ans: convex mirror is used in the view finder of a motor car.
2.8 How does the resistance of semiconductor change with increase fo temperature?
Ans: Resistance decreases with increase of temperature in semiconductor.
2.9 Which type of energy is transferred to electrical energy in a dynamo?
Ans: Mechanical energy is transferred to electrical energy in a dynamo.
2.10 Arrange α, β and γ rays in ascending order of their penetrating power.
Ans: α< β< γ ray.
Or, Which kind of nuclear reaction is the source of the sun’s energy?
Ans: Fusion reaction is the source of the sun’s energy.
2.11 Match the right column with the left column:
| Left Column | Right Column |
|---|---|
| 2.11.1) An alkali metal | K |
| 2.11.2) An element whose anion accelerates rusting of iron | Cl |
| 2.11.3) Extracted from haematite | Fe |
| 2.11.4) Most electronegative element | F |
2.12 What type of chemical bond is present in CaO?
Ans: Ionic bond is present in CaO.
2.13 What is used as cathode to electroplate silver over a copper spoon?
Ans: Copper spoon is used as cathode to electroplate silver over a copper spoon.
Or, Give an example of a compound whose aqueous solution is a weak electrolyte.
Ans: NH3 (Ammonia) is an example of a compound whose aqueous solution is a weak electrolyte.
2.14 During electrolysis which electrode is called a cathode?
Ans: Electrode connected to the negative terminal of the battery is called a cathode.
2.15 State one use of liquid ammonia.
Ans: Liquid ammonia used as Refrigerant.
Or,Write the formula of the precipitate formed when aqueous ammonia solution is added to aqueous solution of aluminium chloride.
Ans: Al(OH)3 is the formula of the precipitate formed when aqueous ammonia solution is added to aqueous solution of aluminium chloride.
2.16 In the laboratory preparation of nitrogen, aqueous solution of which compound is mixed with aqueous solution of ammonium chloride and heated?
Ans: In the laboratory preparation of nitrogen, aqueous solution of Sodium Nitrite (NaNO3) is mixed with aqueous solution of ammonium chloride and heated.
2.17 Write the IUPAC name CH3CH2CHO.
Ans: Propanal is the IUPAC name CH3CH2CHO.
Or,Write the structural formula of positional isomer of CH3CH2CH2OH.
Ans: OH
│
CH3 ━ CH ━ CH3
2.18 Mention one use poly(tetrafluoroethylene).
Ans: Used as a coating in non-stick cook ware due to its hydrophobic and high heat resistant property.
Group-C
3. Answer the following question.
3.1 What is methane hydrate?
Ans: Methane Hydride (4CH4,23H2O is the crystalline solid that consists of a methane molecule surrounded by a cage of interlocking water molecules.
3.2 The pressure of a mixed mass of a gas at temperature of 0°C is doubled while the volume is halved. What will be the final temperature of the gas?
Ans:


Or, Under constant pressure a mixed mass of a gas is heated from 0°C to 546°C. What is the ratio of the final volume of the gas with its initial volume?
Ans:
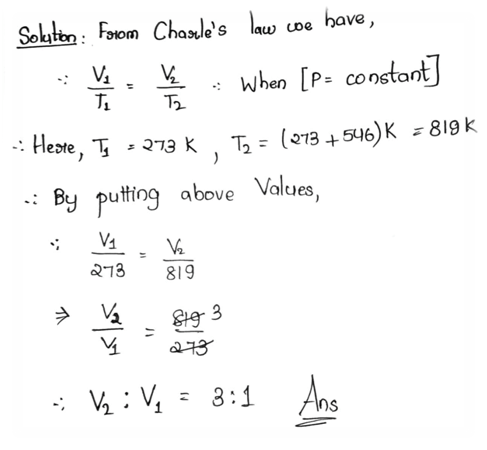
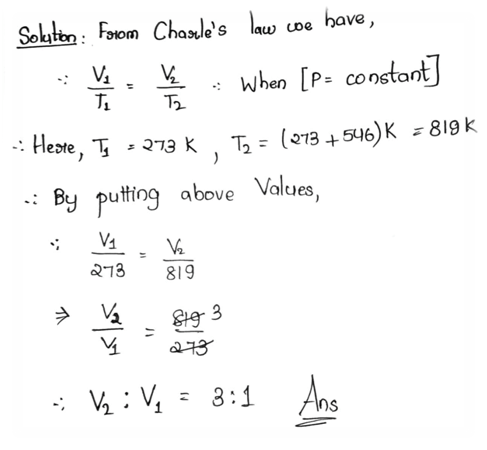
3.3What is meant by the optical centre of a convex lens?
Ans: If a ray of light passes through a Convex lens such that the direction of the emergent rays is parallel to the direction of the incident rays, then the path of the ray of light inside the lens intersects the principal axis at a particular point. This point is called the optical centre of the convex lens.
Or, Why does the earth’s sky appear blue during day time?
Ans: There are so many fine particles and molecules in the atmosphere which are smaller than the wavelength of visible light. These are more effective in scattering light of shorter wavelengths at the blue end of the visible spectrum than at the longer wavelength at the red end. (In fact, wavelength of red light is approximately 1.8 times more than that of the blue light). So, blue light is scattered more as compared to red and reaches our eyes. So, the sky appears blue.
3.4 State Lenz’s law related to electromagnetic induction.
Ans: Lenz law states that, The direction of the induced e.m.f. is such as to oppose the cause of induction or the cause which produces
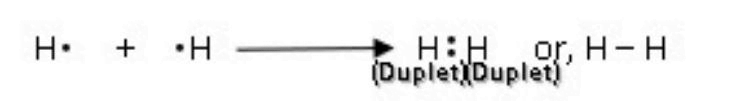
3.5 Write with an example how according to Lewis’s concept a covalent bond is formed.
Ans: The force of attraction which binds atoms of same or different elements by mutual sharing of electron pair(s) is called a covalent bond. In covalent bond the shared electron pair(s) is common to both the atoms.
According to Lewis concept,
Formation of H2 molecule: Atomic number of H is 1. It has only one electron located in K shell. To achieve a stable electronic configuration, which has two electrons in its outermost shell, H-atom needs one more electron. This is achieved by sharing its electron with another H-atom. Thus, two hydrogen atoms share total of two electrons between them, thus forming a hydrogen molecule.

Or,Why the bond in sodium chloride can’t be expressed as Na-Cl?
Ans: For the maximum stability of ionic compounds, cations and anions form crystals by arranging themselves in regular & definite geometrical
pattern so that the coulombic forces of repulsion among the ions of similar charge is minimum. During formation of NaCl crystal, each Na+ ion is surrounded by six neighbouring Clions and each such Cl- ion is similarly surrounded by six neighbouring Nat ions, i.e. each ion lies at the centre of an octahedron and the oppositely charged ions reside at the corners of that octahedron. This type ofcrystal arrangement is known as 6-6 coordination. There is no existence of a separate molecule and the entire crystal exists as a giant molecule.
3.6 Give one example each of a liquid and a solid covalent compound?
Ans: Liquid Covalent compound: Water.
Solid covalent compound: Sugar Crystals.
3.7 Write with balanced chemical equation what happens when H2S gas is passed through an aqueous copper sulphate solution.
Ans: H2S + CuSO4 → Cus + H2SO4
(i) Black precipitate is formed.
(ii) Double Displacement reaction.
3.8 Write down cathode reaction when an aqueous solution of MSO4 (M=metal) is electrolysed. Write with reason whether the reaction is oxidation or reduction.
Ans: Cathode reaction: M2+ + 2e- → M
In Cathode, the cation M2+ takes up the electron to form M. Hence, it is reduction reaction.
Or, Give one use of each of copper and aluminium.
Ans: Use of copper: Copper is extensively used to prepare electrical wire and conductors, switches, pluges etc.
Use of aluminium: Aluminium is used to prepare sheds (roofing), bodies of aeroplane and automobiles, cooking utensils, electrical wire etc.
3.9 What is the condition of substitution reaction of methane with chloride? Write the balanced chemical equation of the first step of the reaction.
Ans: Ethylene molecule has the formula C2H4. Each carbon atom shares two electrons with two hydrogen atoms to form two C-H bonds. Two pairs of electrons are shared between two carbon atoms. So, altogether there are 4 C-H single bonds and one carbon-carbon double bond.
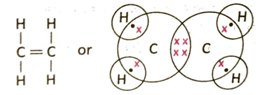

It is evident from the structure that since there existsonedouble bond between the two adjacent carbon atoms in the molecule, hence ethylene is an unsaturated hydrocarbon.
Or,Write with balanced chemical equation what happens when ethanol reacts with metallic sodium.
Ans: To make ethanol unfit for drinking, some amountof poisonous & bad smelling substances such as methanol, pyridine, etc., are added to it. This is then called denatured spirit which is unfit for human consumption but to be used as solvent in industries.
Group-D
4. Answer the following question.
4.1 Establish ideal gas equation on the basis of Boyle’s law, Charles’ law and Avogadro’s law.
Ans: State of any gas can be completely be described by four variables such as pressure (P), Volume (V), Absolute temperature (T) and number of moles (n) of the gas.
According to Boyle’s law, V ∝ 1/P (T and n are constant)
According to Charle’s law, V ∝ T (P and n are constant)
According to Avogadro’s law, V ∝ n (P and T are constant)
By method of compound variation, V ∝nT/p, When P, T and n of the gas vary
Therefore, V ∝RnT/P (R = molar gas constant).
Or, PV = nRT
This equation is called Ideal Gas Equation.
4.2 SO2 required for the industrial production of sulphuric acid is produced by burning iron
pyrites in excess air current. The chemical equation of the reaction is given below: 4FeS2+11O2 → 2Fe2 O3+8SO2
How many grams of FeS2 is required for production of 512 gram of SO2? (Fe=56, S=32,
O=16)
Ans:
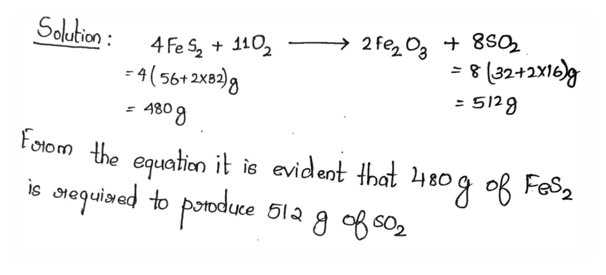
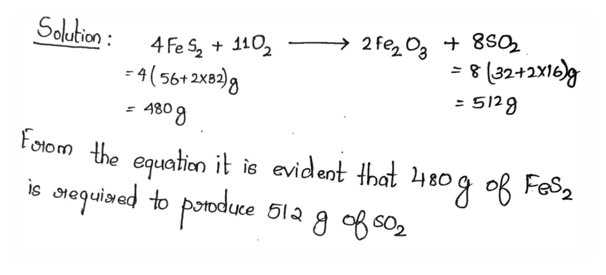
Or, By heating 200 gram of a metal carbonate 112 grams metal oxide and a gaseous compound are processed. Vapour density of the gaseous compound is 22. How many molecules of the gaseous compound is processed in the reaction?
Ans:


4.3 What is thermal conductivity? What is its SI unit?
Ans: Thermal conductivity of a material is the amount of heat which when passes normally in one second through the opposite faces of unit cube (i.e, 1m cube), the difference in temperature between its 2 opposite faces is 10C.
The S.I. unit of thermal conductivity is J.m-1.K-1.s-1.
Or, Define coefficient of surface expansion. Write its SI unit.
Ans: The coefficient of linear expansion of a substance is the change in length relative to its original length at 00Cper unit of temperature.
The S.I. unit of coefficient of linear expansion of a substance is K-1.
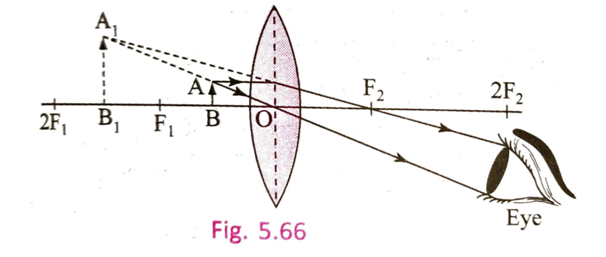
4.4 How can an erect and magnified image be formed with the help of a convex lens? With the help of which type of lens long sightedness can be rectified?
Ans: When the object is placed between focus and optical centre, an erect and magnified image be formed with the help of convex lens.
Long Sightedness is rectified using convex lens.

4.5 If the velocity of light in a medium is 2×10^8 m/s, what will be the refractive index of that medium?
Ans: The R.I of a medium is given by,
μ = Velocity of light in vacumm/Velocity of light in the medium
or, μ = 3 108/2 108
or, μ = 3/2 = 1.5
Or, The refractive index of a medium with respect to air is √2. If the angle of incidence of a ray of light in air is 45° determine the angle of deviation for that ray in case of refraction.
Ans: If ∟i and ∟r be the angle of incidence (in air) & angle of refraction (in the medium) respectively, then according to snell’s law,
Sin i/SinR =μ
Given ∟i = 450 and μ = √2.
By Putting values,
Sin 450 / √2 = ½ =sin 300
∟r = 300.
Since refraction is taking place from rarer to denser medium,
angle of deviation, δ = i – r = 450 – 300 = 150.
4.6 Write Joule’s law related to heating effect of current.
Ans: Joule’s law of heating : If H amount of heat is produced in a conductor of resistance R, when a steady current / is passed through it for time t, then H is
(i) directly proportional to the square of current (H ∝ I2), when R & t are kept constant
(ii) directly proportional to the resistance of the resistor (H ∝ R), when / & t are kept constant
(iii) directly proportional to the time for which the current flows through the resistor (H ∝ t), when / and R are kept constant.
Therefore,
H = kl2 Rt
4.7 Calculate the equivalent resistance when a wire of resistance 10 ohm is divided into equal parts and connected in parallel combination.
Ans:


Or, There are two 60 W lamps and two 80 W fans in a house. The lamps and fans run 5 hours daily. Find out the expense in a month if an unit of electricity costs Rs.4/-. (assume 1 month=30 days)
Ans:
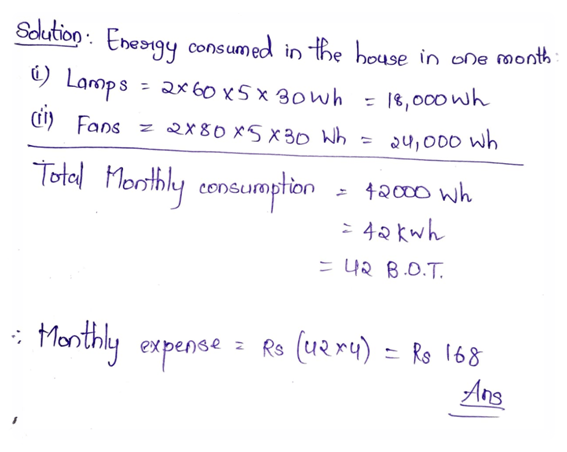
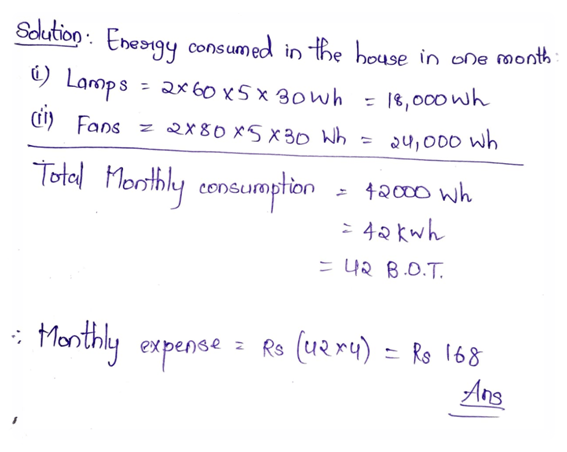
4.8 Compare the charge and ionising power of α and γ rays. Mention one use of radioactivity.
Ans: Use of radioactivity: Radio cobalt is used to kill cancer cells.
4.9What is meant by ionisation energy of an item of an element? Arrange Li,Rb,K and Na in the increasing order of their ionisation energy.
Ans: The energyrequired to remove the most loosely bound electron from an isolated gaseous atom is called ionization energy of an atom of an element.
Rb>K>Na>Li
Or, Mention similarity of properties of hydrogen with one property of group 1 elements and two properties of group 17 elements.
Ans: Similar properties of hydrogen with Group 1 elements:
(i) Valency: Like the alkali metals, hydrogen has only one valence electron and its valency is 1.
Similar properties of hydrogen with group 17 elements:
(i) Electronic configuration: The outer most orbit of both hydrogen and the elements of group 17 have one electron less than the electronic configuration of nearest inert gas. So their valency is 1.
(ii) Non-metallic character and atomicity: Like the elements of group 17, hydrogen is also a non-metal. The molecule of hydrogen is diatomic.
4.10 What are present along with pure alumina in the molten mixture which is electrolysed for the extraction of aluminium by electrolysis? What are used as cathode and anode in this electrolysis?
Ans: Aluminium is obtained by electrolysis of a mixture of pure aluminium (Al2O3) ( 20 parts), cryolite (an ore of aluminium, AlF3.3NaF) ( 60 parts) and fluorspar (CaF2) ( 20 parts) at 9500C.[melting point of pure alumina is very high 20500C].
Cathode: carbon coating in inner wall.
Anode: Carbon rods.
4.11 Write the conditions and balanced chemical equation for the industrial production of
ammonia by Haber’s process.
Ans: Industrially ammonia is synthesized from its constituent elements pure & dry N2& H2 by Haber process. H2 is obtained from synthesis gas and N2 from synthesis gas/air liquefaction process.
N2 + 3H2 = 2NH3
The reaction is carried out at about 5500C and 200 atmospheric pressure in presence of iron catalyst promoted by molybdenum and oxides of potassium and aluminium. The reaction is exothermic, so it should be favoured at lower temperature. But at low temperature, the reaction occurs at a very slow rate, so the reaction is carried out at this optimum temperature. The above reaction is favoured at high pressure and an optimum pressure of 200 atm is employed for production of ammonia by this process.
4.12 The molecular formula of an organic compound is C2H4O2. The compound is soluble in water and on addition of NaHCO3 to the aqueous solution of the compound CO2 is evolved. Identify the organic compound. Write, with condensations and balanced chemical equation, the reaction of the compound with ethanol.
Ans: The compound is acetic acid (CH3COOH). It is soluble in water and on addition of NaHCO3 to its aqueous solution, CO2 is evolved as per the following reaction:
CH3COOH + NaHCO3 → CH3COONa + H2O + CO2
Reaction with NaHCO3: Ethanoic acid reacts with sodium hydrogen carbonate (NaHCO3) to form sodium ethanoate and water along with brisk effervescence of carbon dioxide gas.
CH3COOH + NaHCO3 → CH3COONa + H2O + CO2
Or, Compare three properties of organic and inorganic compounds.4.1 Establish ideal gas equation on the basis of Boyle’s law, Charles’ law and Avogadro’s law.
Ans: