Group-A
1.Multiple choice question.
1.1 Which of the following is not a greenhouse gas?
(a) Methane
(b) water vapour
(c) carbon dioxide
(d) oxygen (Ans)
1.2 Which of the following is the SI unit of pressure?
(a) Nm² (Ans)
(b)Nm-² (Ans)
(c) Nm
(d)N
1.3 Vapour density of a gas is 32. Which of the following is its molecular weight?
(a) 8
(b) 16
(c) 32
(d) 64 (Ans)
1.4 On which of the following does the thermal conductivity of a metal depend?
(a) Temperature
(b) length
(c) nature of the material (Ans)
(d) area of cross section
1.5 What is the relation between the radius of curvature and the focal length of a mirror?
(a) f=2r
(b) f=r/2 (Ans)
(c) f=r/3
(d) f=3/2 r
1.6 Which of the following colours deviates most when white light is refracted by a prism?
(a) red
(b) yellow
(c) violet (Ans)
(d) green
1.7 Which of the following physical quantities represents ampere?
(a) coulomb-second
(b) volt/ohm (Ans)
(c) volt-ohm
(d) ohm/volt
1.8 Resistivity of which of the following decreases with increase in temperature?
(a) conductor
(b) semiconductor (Ans)
(c) super conductor
(d) insulator
1.9 Which of the following is the correct order of ionising power for α,β and γ rays?
(a) α>β>γ
(b) α>γ>β
(c) γ>β>α
(d) β>α>γ (Ans)
1.10 Which of the following has the highest atomic radius?
(a) K (Ans)
(b) H
(c) Li
(d) Na
1.11 In which of the following a covalent bond is present?
(a) Hydrogen chloride (Ans)
(b) sodium chloride
(c) lithium hydride
(d) calcium oxide
1.12 Which of the following is a weak electrolyte in aqueous solution?
(a) CH3-COOH (Ans)
(b) NaOH
(c) H2SO4
(d) NaCl
1.13 Which of the following is used to dry moist ammonia?
(a) conc. H2SO4 (Ans)
(b) P2O5
(c) CaO
(d) CaCl2
1.14 Which of the following is an ore of aluminium?
(a) bauxite (Ans)
(b) Haematite
(c) Malachite
(d) chalcopyrite
1.15 Which of the following is the functional group of aldehydes?
(a) -OH
(b) -CHO (Ans)
(c) >C=O
(d)-COOH
Group-B
2. Answer the following question (alternatives are to be noted).
2.1 In troposphere temperature Decreases with increase in altitude.
Or, Mention one harmful effect of global warming?
Ans: The harmful effect of global warming is Rise in the level of sea water
2.2 Which atom is released from a chlorofluorocarbon under the influence of UV rays, decomposes ozone oxygen?
Ans: Chlorine is released from a chlorofluorocarbon under the influence of UV rays, decomposes ozone oxygen.
2.3 Write whether the following statement is true or false
Under the same condition of temperature and pressure equal volumes of CO2 and N2 gas contain different number of molecules. False
2.4 Write down the relation between the pressure and volume of a given mass of a gas according to Boyle’s law when temperature remains constant.
Ans: For a given mass of a gas at constanttemperature, V ∝ 1/P or V= K/P(K =proportionality constant) or PV = K = constant.
2.5 Between apparent and real expansion coefficient of a liquid which one is its own characteristic?
Ans: Real expansion coefficient of liquid is its own characteristic.
Or, what is the SI unit of thermal conductivity?
Ans: SI unit of thermal conductivity is W/mk.
2.6 Give a natural example of dispersion of light?
Ans: Rainbow is an example of natural dispersion of light.
2.7 Which type of lens can rectify myopic vision?
Ans: Concave lens can rectify myopic vision.
2.8 What will be the change in the motion of a Barlow’s wheel if the direction of the current is reversed?
Ans: If the direction of the current is reversed in Barlow’s wheel then the motion will also be reversed.
2.9 How much work has to be done when 1 coulomb of charge is taken against a potential difference of 1 volt.
Ans: 1J work has to be done when 1 coulomb of charge is taken against a potential difference of 1 volt.
2.10 From which part of a radioactive atom is β-particle emitted?
Ans: From nucleus of a radioactive atom is β-particle emitted.
2.11 Match the right column with the left column:
| Right Column | Left Column |
|---|---|
| 2.11.1) Transition element | Nickel |
| 2.11.2) Transuranium element | Plutonium |
| 2.11.3) Reduces Fe2O3 at high temperature in thermite process | Aluminium |
| 2.11.4) Present in alloy bell metal | Tin |
2.12 Draw Lewis dot diagram of F2 molecule. (F=9)
Ans: Electronic configuration of F : K = 2, L = 7
By sharing one of its valence shell electrons with other, two F atoms form a covalent single bond between them to form the F, molecule. Thus each atom attains the configuration of inert gas Ne (2, 8).

2.13 Why the electrical conductivity of the solution produced by adding little sulphuric acid into pure water is more than the electrical conductivity of pure water?
Ans: Pure water is a bad conductor of electricity i.e ., a very weak electrolyte which dissociates to a very small extent as,H 2O = > H+ + OH–.
The bare proton (H+) immediately associates with a molecule of H2O to form the hydronium ion, H3O+ : H+ + H2O => H3O+. But water becomes a good conductor of electricity and can be electrolysed when a very small amount of a strong acid (e.g. HCI or H2SO4) or a strong alkali (e.g ., NaOH, KOH) is added to it.
Or, what are the carriers of electricity in the solution during the electrolysis of an aqueous solution of an electrolyte?
Ans: Ions are the carriers of electricity in the solution during the electrolysis of an aqueous solution of an electrolyte.
2.14 Which ion migrates towards the cathode during the electrolysis of aqueous solution of CuSO4 using Cu-electrodes?
Ans: When an aqueous solution of CuSO4 is electrolysed using Cu-electrodes, both Cu+2 and H+ ions migrate towards the cathode, where Cu+2 ions are discharged in preference to H+ Ions.
2.15 Write down the formula of black precipitate formed when H2S gas is passed through an aqueous solution of lead nitrate?
Ans: The formula of black precipitate formed when H2S gas is passed through an aqueous solution of lead nitrate is lead sulphide (PbS)
Or, what change of colour is observed when H2S gas is passed through an acidified aqueous solution of potassium dichromate?
Ans: When a orange colour acidified solution of potassium dichromate reacts with H2S, it is reduced to green coloured chromium sulphate [Cr2(SO4)3].
2.16 Two substances are used in the production of urea. One is ammonia, what is the other?
Ans: The other one is CO2 (carbon- dioxide).
2.17 Mention one use of poly vinyl chloride?
Ans: Poly vinyl chloride is used to manufacture fire resistant floor tiles and other interior housing materials.
2.18 Write down the IUPAC name of CH3CH2OH?
Ans: The IUPAC name of CH3CH2OH is Propanol.
or,
Ans: CH3 – O – CH3 (dimethyl ether)
Group-C
3. Answer the following questions.
3.1 What is meant by calorific value of a fuel? Between coal and diesel which has greater calorific value?
Ans: Calorific value of a fuel may be defined as the amount of heat liberated in calories or joules by the complete combustion of unit weight of a combustible material with oxygen and the condensation of the products to the desired temperature. It is generally expressed in units of kilo calorie per kg or kilojoule per kg.
Diesel (45 KJ/g) has a greater calorific value than coal (25 to 35 Kj/g).
3.2 If the volume of a given mass of a gas at STP is 52m³ what will be its volume at the pressure of 104 cm Hg temperature remaining constant?
Ans: Here, P1 = 76cm Hg (STP), V1 = 52m3, P2=104cm Hg and V2= ?
At constant temperature,
P1V1 = P2V2
Or, 76 52 = 104 V2
Or, V2 = 76 52/104=38m3
Or, what will be the volume of 8g of H2 gas (H=1) at the pressure of 4 atmosphere and temperature of 300K?
(R=0.082 litre atmosphere 〖mol〗^(-1) K^(-1))
Ans: OR,
Here, P = 4 atm, V = ?
n (no. of moles of H2 gas) = 8/2 = 4
R = 0.082 litre – atm .mol-1K-1 and T = 300K.
From the equation of state we have ,
PV =nRT
Or, V = nRT/P = 4 0.082 300/4 = 24.6L
3.3 How does the refractive index of a medium depends on the velocity of light?
Ans: The absolute refractive index of a medium is defined as the ratio of the speed of light in vacuum to the speed of light in that medium. So, absolute refractive index of a medium (u),
speed of light in vacuum u=speed of light in that medium
This means that higher the speed of light in a medium, lower will be the absolute refractive index of that medium. Light travels faster in an optically less dense medium than inan optically more dense medium.
Or, what kind of mirror is used in the headlight of a car and why?
Ans: Concave mirror is used as reflector is used as a reflector in the headlight of cars.
When a source of light is placed at the principal focus of a concave mirror, it produces a strong parallel beam of light. thus it used as a reflector in automobile’s headlights and search lights.
3.4 Mention two advantages of using a CFL lamp over a incandescent lamp?
Ans: Advantages of CFL over incandescent lamps are listed below:
(i) CFL consumes less electrical energy.
(ii) CFL’shave a life span is almost five times of an incandescent lamp. Therefore the higher purchase price compared to that of an incandescent lamp is compensated for by the saving in energy consumption over an enhanced life span.
3.5 An aqueous solution of sugar cannot conduct electricity, but an aqueous solution of sodium chloride can.Why?
Ans: Sugarorsucrose, a disaccharide of glucose and fructose, is a covalent compound. It does not possess negatively or positively charged ions and so, it does not conduct electricity in dissolved state. In aqueous solution of sodium chloride, the ions (Na+ and Cl– ) become free from the crystal lattice and hence they conduct electricity on account of their mobility.
Or, why is it appropriate to use the term formula weight instead of molecular weight in case of sodium chloride?
Ans: A conglomeration of countless number of cations and anions leads to the formation of crystals of an ionic compound like sodium chloride. The cations (Na+) and anions (CI–) form crystals by arranging themselves in a regular octahedron to form the NaCl crystal. Hence, there is no existence of a separate molecule and the entire crystal exists as a giant molecule. Thus formula weight is more appropriate to be used for NaCl instead of molecular weight.
3.6 Compare the following two properties of naphthalene and sodium chloride: melting point and solubility in water.
Ans:
3.7 Name a gas which can be prepared in Kipp’s apparatus. Write the balanced chemical equation of the reaction for the preparation of the gas.
Ans: H2S can be readily and intermittently prepared in kipp’sappratutus.
H2S is prepared by the action of dilute sulphuric acid on ferrous sulphide.
FeS + H2SO4 = FeSO4 + H2S
3.8 How is metallic zinc is obtained from zinc oxide? Write reaction with balanced chemical equation.
Ans: Zinc oxide is reduced to metallic zinc by heating it with carbon. Here carbon plays a role of reducing agent .
ZnO + C →Zn+CO
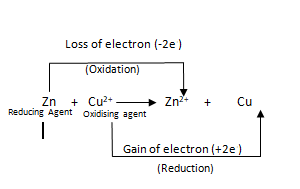
Or, what happens when a piece of zinc is added to an aqueous solution of CuSO4? Show with the help of electronic theory that this is an oxidation-reduction reaction.
Ans: When a strip of silvery white zinc metal is immersed in a blue coloured solution of copper sulphate (CuSO4), then copper sulphate is reduced to metallic copper and zinc is oxidized to zinc sulphate (ZnSO4). Hence the blue colour of the solution gradually fades and the metal is covered by a reddish brown coating of metallic copper.
Zn + CuSO4→Cu+ZnSO4
The reaction can be represented in ionic form as follows:
Zn + Cu+2→ Zn+2 + Cu
Zn atom loses two electrons to form Zn+2 ion (oxidation). On the other hand, Cu+2 ion accepts two electrons to produce Cu atom (reduction).Thus, this is an oxidation – reduction reaction.

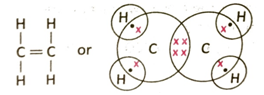
3.9 Show with the help of structural formula of ethylene that it is an unsaturated hydrocarbon?
Ans: Ethylene molecule has the formula C2H4. Each carbon atom shares two electrons with two hydrogen atoms to form two C-H bonds. Two pairs of electrons are shared between two carbon atoms. So, altogether there are 4 C-H single bonds and one carbon-carbon double bond.
It is evident from the structure that since there existsonedouble bond between the two adjacent carbon atoms in the molecule, hence ethylene is an unsaturated hydrocarbon.

Or, what is denatured spirit?
Ans: To make ethanol unfit for drinking, some amountof poisonous & bad smelling substances such as methanol, pyridine, etc., are added to it. This is then called denatured spirit which is unfit for human consumption but to be used as solvent in industries.
Group-D
4. Answer the following questions.
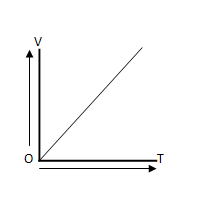
4.1 State Charles’ law related to gases and represent it graphically (V vs I)?
Ans: At constant temperature, the volume of a given mass of gas is increased or decreased by 1/273 parts of its volume at 00C for every 10 rise or fall in temperature.
Where, V= volume of the gas , T = temperature of the gas, and O = point of origin.

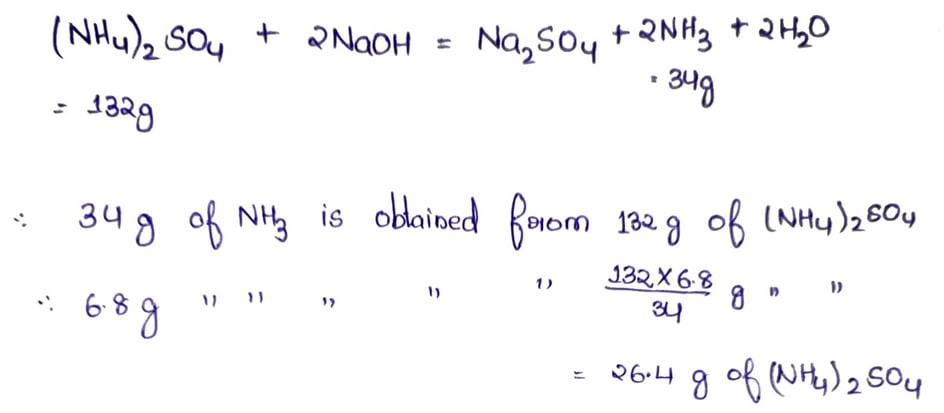
4.2 How many grams of ammonium sulphate will be required to prepare 6.8 g of ammonia by heating ammonium sulphate with caustic soda solution? (H=1, N=14, O=16, S=32)
Ans:

Or, CaCl2, CO2 and H2O are produced by the reaction of dilute HCl with CaCO3. How many grams of HCl will be required to prepare 55.5 g of CaCl2, 22.0 g of CO2 and 9.0 g of H2O from 50.0 g of CaCO3? What is the number of moles of HCl required? (H=1, Cl=35.5)
Ans:


4.3 What is the volume expansion coefficient of a gas at fixed pressure? What is its value?
Ans: At constant pressure, the volume of a given mass of an ideal gas increases by a constant fraction,1/273 of its volume at 00C for each degree Celsius rise in temperature. This constant fraction is the coefficient of expansion of ideal gas at constant pressure.
Thus, the volume expansion coefficient of a gas at fixed pressure (γ) = 1/273
or, Mention two similarities between heat conduction and electrical conduction. Mention a non-metal having high thermal conductivity
Ans: Diamond, an allotrope of carbon, has a high value of thermal conductivity (2200 Wm-1K-1).
4.4 State the two laws of refraction of light. Write also the mathematical form of the second law.
Ans: The following are the two laws of refraction:
i)The incident ray , the refracted ray and the normal at the point of incidence on the surface of separation lie in the same plane.
ii)The ratio of the sine of the angle of incidence to the sine of the angle of refraction for any two media is constant for the light of a particular colour and for a given pair of materials. This law is known as Snell’s law after the name of the Dutch scientist Willebrad Snell who first proposed this relation.
The ratio is called the refractive index (or in short R. I) of the second medium with respect to the first medium. It is usually represented as 1μ2.
So, sin i/sin r = 1μ2
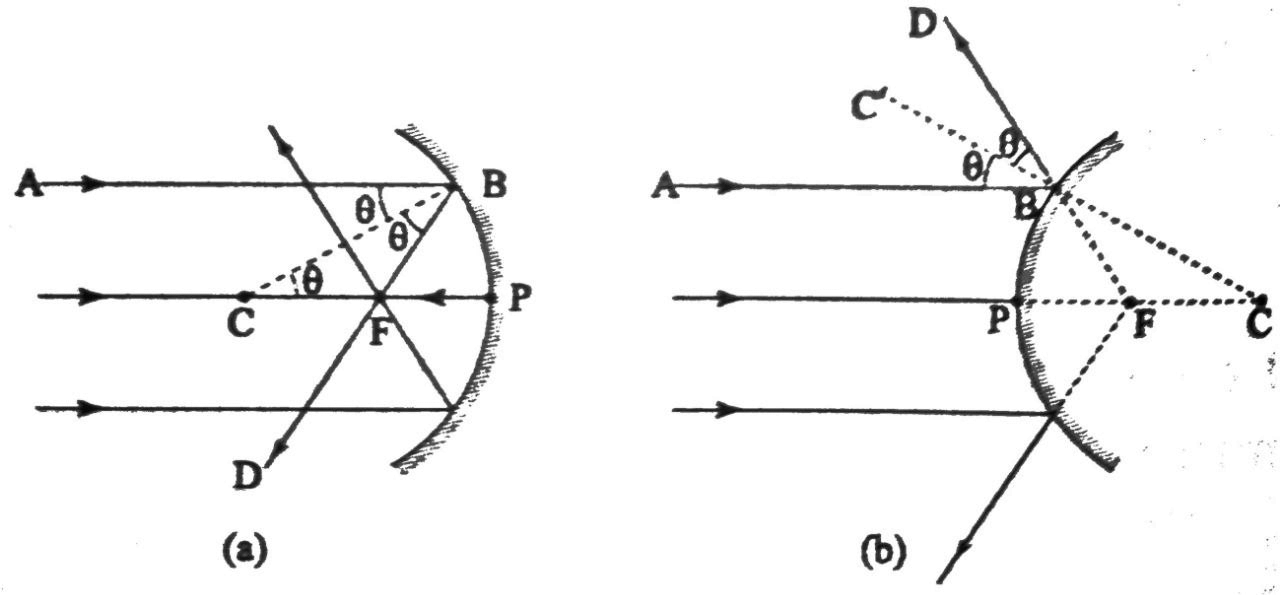
4.5 In case of a concave mirror find the relationship between focal length and radius of curvature.
Ans: A relation between the radius of curvature (r) and the focal length (f) of a spherical mirror can be established for paraxial rays.
In Fig. (a) and (b), F is the focus, C is the centre of curvature and P is the pole of the spherical mirror. PF is the focal length (f) of the mirror. PC is the radius of curvature (r). A paraxial ray of light AB, parallel to the principal axis is
Incident on Band is reflected along BD. For the concave mirror (a) it passes through the focus(F)and for the convex mirror (b) it appears to diverge from F. BC is normal to the tangentdrawn at B, the point of incidence, on the spherical mirror.
Let us now do some simple geometry and derive the relation for concave mirror.
As per the laws of reflection, the angle of incidence, LABC = angle of reflection, LCBF.
Since, AB is parallel to principal axis CP, LABC= LBCF (since they are alternate angles)
Hence, LCBF = LBCF
This means, CFB is an isosceles triangle. Hence, CF = FB
Again if we assume that the apearture of the mirror is small and the rays are paraxial, we can say that FB ~
FP i.e. CFFP.
We have just derived that, CF = FP or, CF + PF = PF+ PF or, PC = 2 PF or, r = 2f or, f = r/ 2.
Thus, we can say that focal length of a spherical mirror is half of its radius of curvature.

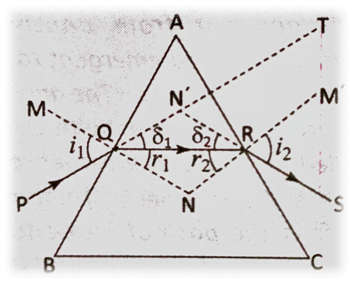
Or, A ray of light passes through a prism. Show that the angle of deviation (δ=i_1+i_2-A)
Ans: The figure, the angle of deviation is LTN’S. Let, LTN’S = δ. In case of refraction by first refracting surface AB, the refracted ray suffers a deviation by an angle, LN’QR and in case of refraction by the second refracting surface AC, the refracted ray suffers a deviation by an angle, LN’RQ. Let, LN’QR = δ1 and LN’RQ = δ2.
It is evident that for QN’R, LN’QR + LN’RQ + LQN’R = 1800
or, LN’QR + LN’RQ = 1800 – LQN’R ……….(1)
Or, δ1 + δ2 = 1800
Again,LTN’S + LQN’R = 1800
or, δ = 1800-LQN’R ……………(2)
So, comparing equation (1) and (2)
δ = δ1 + δ2 …………………….(3)
But,δ1 = i1–r1andδ2 = i2 – r2
So, equation (3) becomes, δ = (i1 – r1) + (i2 – r2 ) or,δ =(i1 + r1) + (i2 + r2 ) ………………………………….. (4)
In the quadrilateral AQNR, LAQN = LNRA =900
So, LQAR + LQNR = 1800
But, LQAR = angle of the prism = A.
Hence, LQNR = 1800– A ……………………………………(5)
But in the triangle QNR, LRQN + LQRN + LONR = 1800
or, r1 + r2 +LQNR = 1800
or, r1 + r2 + (1800– A) = 1800or, r1 + r2 = A ……………………………..(6)
So, substituting the value of (r1 + r2 ) from equation (6) in equation (4),
we get, δ = (i1+ i2) – A
Hence,Proved

4.6 Write Faraday’s law of electromagnetic induction. What is meant by alternating current?
Ans: Faraday law of electromagetic induction:
(i) whenever there is change in the magnetic flux linked with a coil, an emf is induced, which lasts as long as the change in the magnetic flux linked with coil lasts.
(ii) The magnitude of the induced emf is directly propotional to the rate of change of magnetic flux linked with the coil.
Altenating current(A.C) is anelectric current whose magnitude changes with time and direction reverses periodically.
4.7 An electric bulb of resistance 440ohm is connected to 220-volt mains for 10 hours. Find the amount of electrical energy consumed in BOT unit.
Ans:
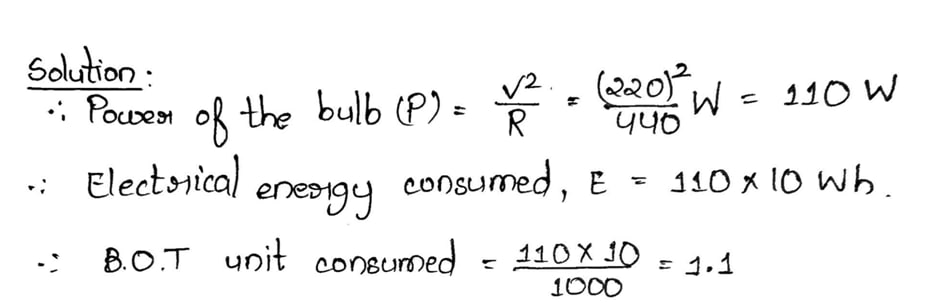
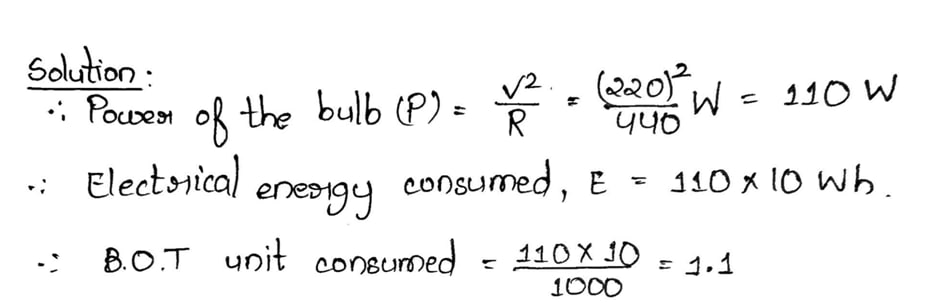
Or, A metal wire of resistance 6 ohm is stretched so that its length become double of ts original length. What would be the final resistance?
Ans:


4.8 What is meant by mass defect? What is the source of energy released in nuclear fusion?
Ans: The difference between the actual mass of a nucleus and its expected mass (as calculated by adding masses of all nucleons) is called mass defect (m) of the nucleus.
m = Z.mp + (A-Z) mn – MN
In nuclear fusion it is found that the heavier nucleus has a mass less than that of the sum of the masses of the reacting lighter nuclei. Therefore, due to conversion of mass defect into energy, fusion is accompanied with release of high amount of energy.
4.9 write with an example what is meant by periodic property of elements? Mention a property which is not periodic.
Ans: According to modern periodic law, there is a repetition of properties of the elements after certain regular intervals when they are arrangedinthe order of their increasing atomic number.
With successive increase of atomic number, there occurs a repetition of similar outermost shell electronic configuration or valence shell electronic configuration after certain regular intervals. Hence by correlation of these two facts it can be said that the periodicity in properties of elements is due to the recurrence of similar outermost shell electronic configuration after certain regular intervals when the elements are arranged in the order of increasing atomic number. The properties of the elements which are directly or indirectly related to their electronic configurations and show a regular gradation when someone moves across a period or descends down a group in the periodic table are called periodic properties.
Examples of periodic property: atomic radii, ionization energy, metallic character, valency etc.
The periodicity of property of alkali metals may be shown as :
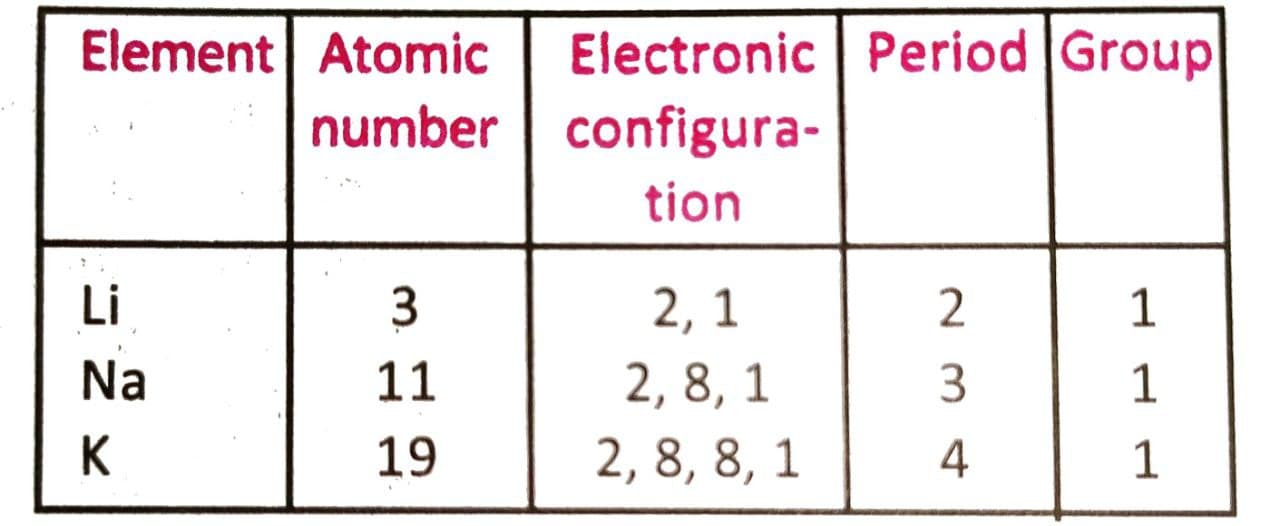

These elements exhibit similar chemical properties since they have similarity in their valence shell electronic configurations and are thus placed in group 1 of successive periods (with gradual increase of atomic number). Radioactivity is not a periodic property of elements since it depends on the ratio between the number of neutrons and protons present in the nucleus of the atom.
Or, What is meant by electronegativity of an element? How does electrone